
James Knierim
Professor of Neuroscience
Contact Information
- jknierim@jhu.edu
- 403 Macaulay Hall/337 Krieger Hall
- Office: 410-516-5170 |
- Group/Lab Website
Research Interests: Behavioral Neurophysiology of the Hippocampal Formation
Education: PhD, California Institute of Technology
Dr. James Knierim is a professor of neuroscience at the Johns Hopkins University School of Medicine. His research focuses on the neurophysiology of memory in the hippocampal formation. Dr. Knierim is a researcher at the Zanvyl Krieger Mind/Brain Institute at Johns Hopkins.
His work has investigated how the zero-gravity environment of NASA's Space Shuttle affects spatial orientation; how the sense of direction (your "internal compass") affects spatial perceptions; and how objects and landmarks become incorporated into the brain's "cognitive map" of an environment in ways that are crucial for the normal formation of long-term memories. Currently, Dr. Knierim is focused on understanding the information processing that occurs in different stages of the hippocampus, from the input representations of the entorhinal cortex through the different subregions within the hippocampus.
After graduating from Haverford College with a BA in psychology, he obtained his PhD in neurobiology at California Institute of Technology, where he studied the primate visual system with David Van Essen. He then did a postdoctoral fellowship with Bruce McNaughton at the University of Arizona, where he studied the spatial firing characteristics of place cells and head direction cells of the rat hippocampus and limbic system. In 1998, he started his own laboratory in the Department of Neurobiology and Anatomy at the University of Texas Medical School at Houston. He joined the Johns Hopkins faculty in 2009.
Behavioral Neurophysiology of the Hippocampal Formation
Work in our laboratory attempts to understand the flow of information through the hippocampal formation and the computations performed by the various subfields of the hippocampus and its inputs from the entorhinal cortex. To address these issues, we use multi-electrode arrays to record the extracellular action potentials from scores of well-isolated hippocampal neurons in freely moving rats. These neurons have the fascinating property of being selectively active when the rat occupies restricted locations in its environment. They are termed "place cells," and it has been suggested that these cells form a cognitive map of the environment (O'Keefe and Nadel, The hippocampus as a cognitive map). The animal uses this map to navigate efficiently in its environment and to learn and remember important locations. It is also hypothesized that these cells play a major role in the formation of episodic (autobiographical) memories. Place cells thus constitute a tremendous opportunity to investigate the mechanisms by which the brain transforms sensory input into an internal, cognitive representation of the world "out there" and then uses this representation as the framework that organizes and stores memories of past events.
Example of Research
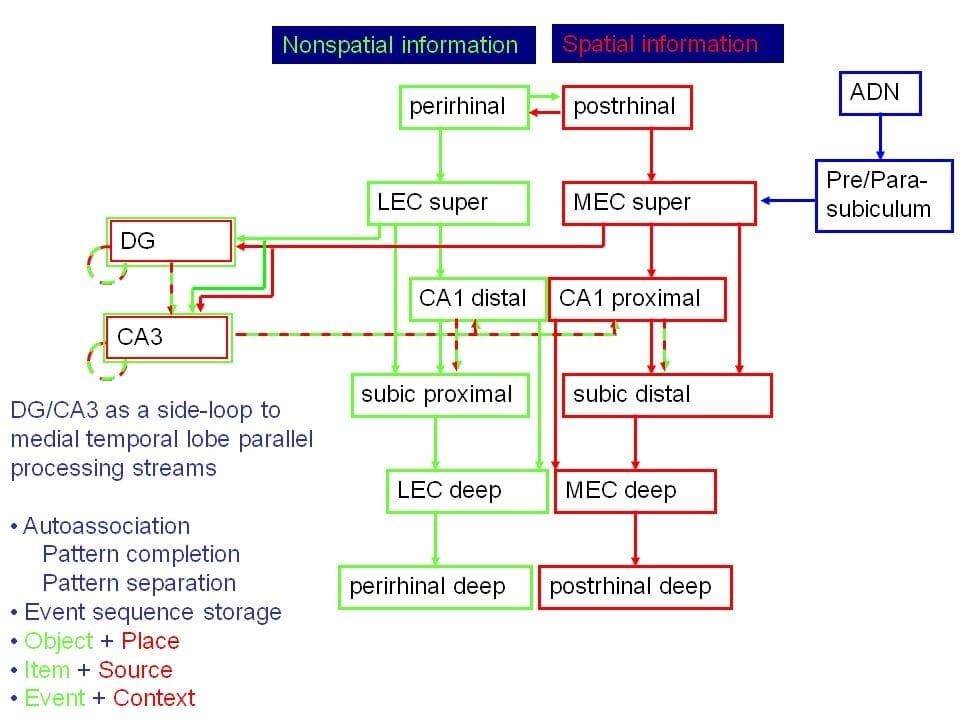 Parallel streams of processing through the hippocampus. The medial entorhinal cortex provides spatial input through the firing of grid cells. The lateral entorhinal cortex provides object and local-cue information. The dentate gyrus and CA3 regions provide a processing side-loop that is essential for such memory functions as autoassociation and object + place (or item + context) configurations, which may be the critical framework for storing episodic memories.
Parallel streams of processing through the hippocampus. The medial entorhinal cortex provides spatial input through the firing of grid cells. The lateral entorhinal cortex provides object and local-cue information. The dentate gyrus and CA3 regions provide a processing side-loop that is essential for such memory functions as autoassociation and object + place (or item + context) configurations, which may be the critical framework for storing episodic memories.
Research Papers
- Wang C, Lee H, Rao G, Doreswamy Y, Savelli F, Knierim JJ. Superficial-layer vs. deep-layer lateral entorhinal cortex: coding of allocentric space, egocentric space, speed, boundaries, and corners. Hippocampus 2023; 33:448-464.
- Kim SK, GoodSmith D, Temme SJ, Moriya F, Ming G-L, Christian KM*, Song H*, Knierim JJ* Global remapping in granule cells and mossy cells of the mouse dentate gyrus. Cell Reports 2023; 42(4) (online ahead of print).
- Madhav MS, Jayakumar RP, Lashkari SG, Savelli F, Blair HT, Knierim JJ, Cowan NJ. The Dome: A virtual reality apparatus for freely locomoting rodents. J. Neurosci. Methods 2022; Volume 368:109336. doi: 10.1016/j.jneumeth.2021.109336.
- Vagvolgyi BP, Jayakumar RP, Madhav MS, Knierim JJ, Cowan NJ. Wide-angle, monocular head tracking using passive markers. J. Neurosci. Methods 2022 Volume 368: 109453. doi: 10.1016/j.jneumeth.2021.109453.
- GoodSmith D, Kim SH, Puliyadi V, Ming G-L, Song H, Knierim JJ, Christian KM. Flexible encoding of objects and space in single cells of the dentate gyrus. Curr. Biol. 2022; 32:1088-1101.
- Lee H, Wang Z, Tillekeratne A, Lukish N, Puliyadi V, Zeger S, Gallagher M, Knierim JJ. Loss of functional heterogeneity along the CA3 transverse axis in aging. Curr. Biol. 2022; 32:2681-2693.
- Lee H, Wang Z, Zeger SL, Gallagher M, Knierim JJ. Heterogeneity of age-related neural hyperactivity along the CA3 transverse axis. J. Neurosci. 2021; 41:663-673.
- Rao G, Lee H, Gallagher M, Knierim JJ. Decreased investigatory head scanning during exploration in learning-impaired, aged rats. Neurobiol. Aging 2020; 98:1-9. doi: 10.1016/j.neurobiolaging.2020.10.008. Online ahead of print.
-
Wang C, Monaco JD, Knierim JJ. Hippocampal place cells encode local surface texture boundaries. Curr. Biol. 2020; 30:1397-1409.
-
GoodSmith D, Lee H, Neunuebel JP, Song H, Knierim JJ. Dentate gyrus mossy cells share a role in pattern separation with dentate granule cells and proximal CA3 pyramidal cells. J. Neurosci. 2019; 39:9570-9584.
- Jayakumar RP, Madhav MS, Savelli F, Blair HT, Cowan, NJ, Knierim JJ. Recalibration of path integration in hippocampal place cells. Nature 2019;566:533-537.
- Branch A, Monasterio A, Blair G, Knierim JJ, Gallagher M, Haberman RP. Aged rats with preserved memory dynamically recruit hippocampal inhibition in a local/global cue mismatch environment. Neurobiol Aging 2019; 76:151-161.
- Wang C, Chen X, Lee H, Deshmukh SS, Yoganarasimha D, Savelli F, Knierim JJ. Egocentric coding of external items in the Lateral Entorhinal Cortex. Science 2018; 362:945-949.
- Tsao A, Sugar J, Lu L, Wang C, Knierim JJ, Moser M-B, Moser EI. Integrating time from experience in lateral entorhinal cortex. Nature 2018; 561:57-62.
- Savelli F, Luck JD, Knierim JJ. Framing of grid cells within and beyond navigation boundaries. eLife 2017 http://dx.doi.org/10.7554/eLife.21354 .
- GoodSmith D, Chen X, Wang C, Kim SH, Song H, Burgalossi A, Christian KM, Knierim JJ. Spatial representations of granule cells and mossy cells of the dentate gyrus. Neuron 2017; 93:677-690.
- Lee H, Wang C, Deshmukh SS, Knierim JJ. Neural population evidence of functional heterogeneity along the CA3 transverse axis: Pattern completion vs. pattern separation. Neuron 2015; 87:1093-1105.
- Monaco JD, Rao G, Roth ED, Knierim JJ. Attentive scanning behavior drives one-trial potentiation of hippocampal place fields. Nature Neurosci. 2014; 17:725-731.
- Neunuebel JP, Knierim JJ. CA3 retrieves coherent spatial representations from degraded dentate gyrus input: Direct evidence for pattern completion in CA3 and pattern separation in the dentate gyrus. Neuron 2014; 81:416-427.
- Neunuebel JP, Yoganarasimha D, Rao G, Knierim JJ. Conflicts between local and global spatial frameworks dissociate neural representations of the lateral and medial entorhinal cortex. J. Neurosci. 2013; 33:9246-9258.
- Deshmukh SS, Knierim JJ. Influence of local objects on hippocampal representations: landmark vectors and memory. Hippocampus 2013; 23:253-267.
- Deshmukh SS, Johnson JL, Knierim JJ. Perirhinal cortex represents nonspatial, but not spatial, information in rats foraging in the presence of objects: Comparison with lateral entorhinal cortex. Hippocampus 2012; 22:2045-2058.
- Roth ED, Yu X, Rao G, Knierim JJ. Functional differences in the backward shifts of CA1 and CA3 place fields in novel and familiar environments. PLoS ONE. 2012; 7(4): e36035 (doi:10.1371/journal.pone.0036035).
- Neunuebel JP, Knierim JJ. Spatial firing correlates of physiologically distinct cell types of the rat dentate gyrus. J. Neurosci. 2012;32:3848-3858.
- Deshmukh SS, Knierim JJ. Representation of non-spatial and spatial information in the lateral entorhinal cortex. Front. Behav. Neurosci. 2011 5:69. (doi: 10.3389/fnbeh.2011.00069).
- Monaco JD, Knierim JJ, Zhang K. Sensory feedback, error correction, and remapping in a multiple oscillator model of place cell activity. Front. Comput. Neurosci. 2011 5:39 (doi:10.3389/fncom.2011.00039).
- Yoganarasimha D, Rao G, Knierim JJ. Lateral entorhinal neurons are not spatially selective in cue-rich environments. Hippocampus 2011 21:1363-74.
- Deshmukh S, Yoganarasimha D, Voicu H, Knierim JJ. Theta modulation in the medial and the lateral entorhinal cortex. J. Neurophysiol. 2010; 104:994-1006.
- Savelli F, Knierim JJ. Hebbian analysis of the transformation of medial entorhinal grid-cell inputs to hippocampal place fields. J. Neurophysiol. 2010; 103:3167-3183.
- Savelli F, Yoganarasimha D, Knierim JJ. Influence of boundary removal on the spatial representations of the medial entorhinal cortex. Hippocampus 2008;18:1270-1282.
- Yu X, Shouval HS, Knierim JJ. A biophysical model of synaptic plasticity and metaplasticity can account for the dynamics of the backward shift of hippocampal place fields. J. Neurophysiol. 2008; 100:983-992.
- Siegel JJ, Neunuebel JP, Rao G, Knierim JJ. Dominance of the proximal coordinate frame in determining the locations of hippocampal place cell activity during navigation. J. Neurophysiol. 2008; 99:60-76.
- van der Meer MAA, Knierim JJ, Yoganarasimha D, Wood ER, van Rossum MCW. Anticipation in the rodent head direction cell system can be explained by an interaction of head movements and vestibular firing properties. J. Neurophysiol. 2007; 98:1883-1897.
- Knierim JJ, Lee I, Hargreaves EL. Hippocampal place cells: Parallel input streams, subregional processing, and implications for episodic memory. Hippocampus 2006; 16:755-764.
- Yu X, Yoganarasimha D, Knierim JJ. Backward shift of head direction tuning curves of the anterior thalamus: Comparison with CA1 place fields. Neuron 2006; 52:717-729.
- Yoganarasimha D, Yu X, Knierim JJ. Head direction cell representations maintain internal coherence during conflicting proximal and distal cue rotations: Comparison with hippocampal place cells. J. Neurosci. 2006; 26:622-631.
- Hargreaves EL, Rao G, Lee I, Knierim JJ. Major dissociation between medial and lateral entorhinal input to dorsal hippocampus. Science 2005; 308:1792-1794.
- Yoganarasimha D, Knierim JJ. Coupling between place cells and head direction cells during relative translations and rotations of distal landmarks. Exp. Brain Res. 2005; 160:344-359.
- Guzowski JF, Knierim JJ, Moser EI. Ensemble dynamics of hippocampal subregions CA3 and CA1. Neuron 2004; 44:581-584.
- Lee I, Yoganarasimha D, Rao G, Knierim JJ. Comparison of population coherence of place cells in hippocampal subfields CA1 and CA3. Nature 2004; 430:456-459.
- Lee I, Rao G, Knierim JJ. A double dissociation between hippocampal subfields: Differential time course of CA3 and CA1 place cells for processing changed environments. Neuron 2004; 42:803-815.
- Knierim JJ, Rao G. Distal landmarks and hippocampal place cells: Effects of relative translation vs. rotation. Hippocampus 2003; 13:604-617.
- Knierim JJ. Dynamic interactions between local surface cues, distal landmarks, and intrinsic circuitry in hippocampal place cells. J. Neurosci. 2002; 22:6254-6264.
